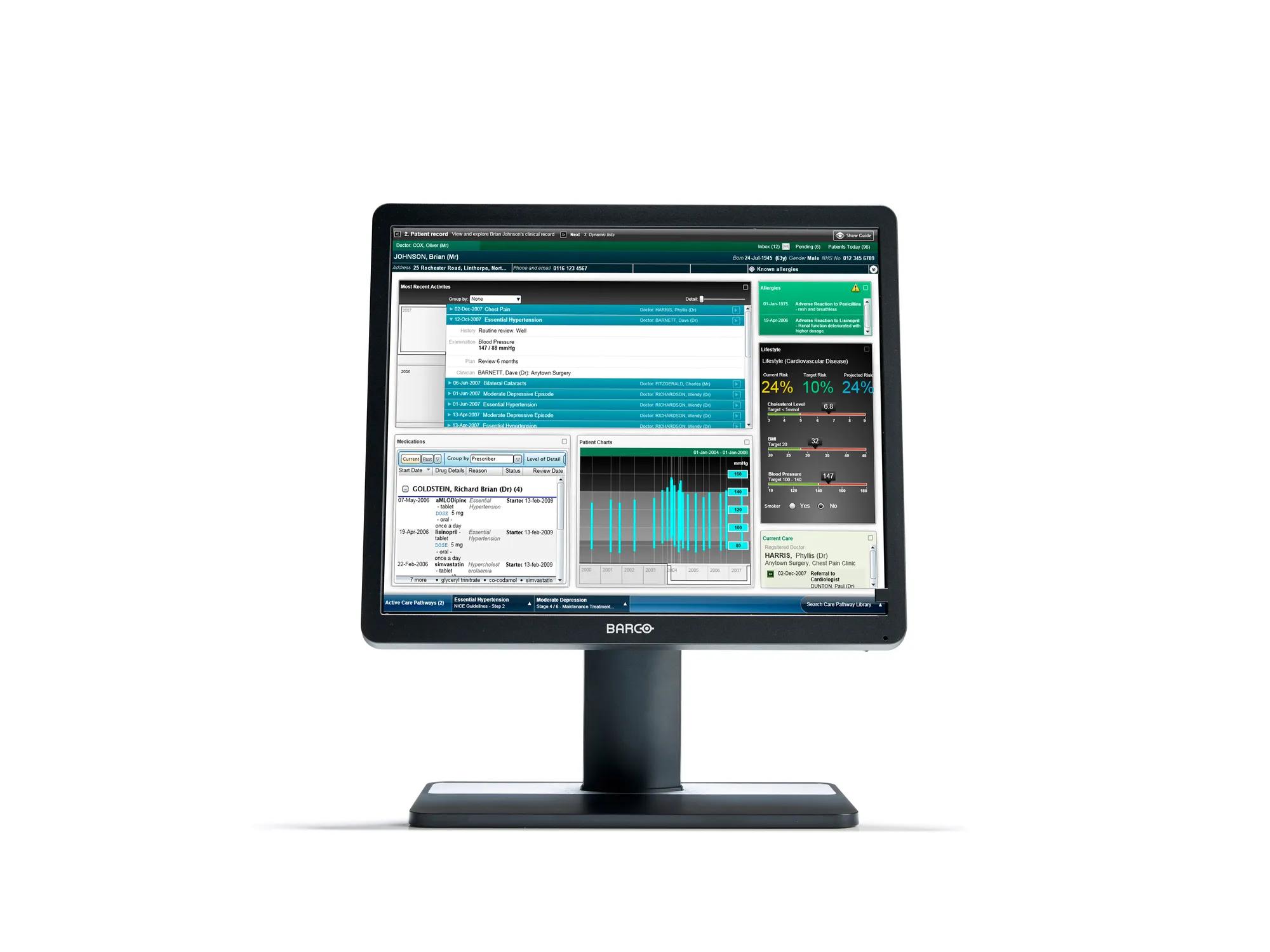
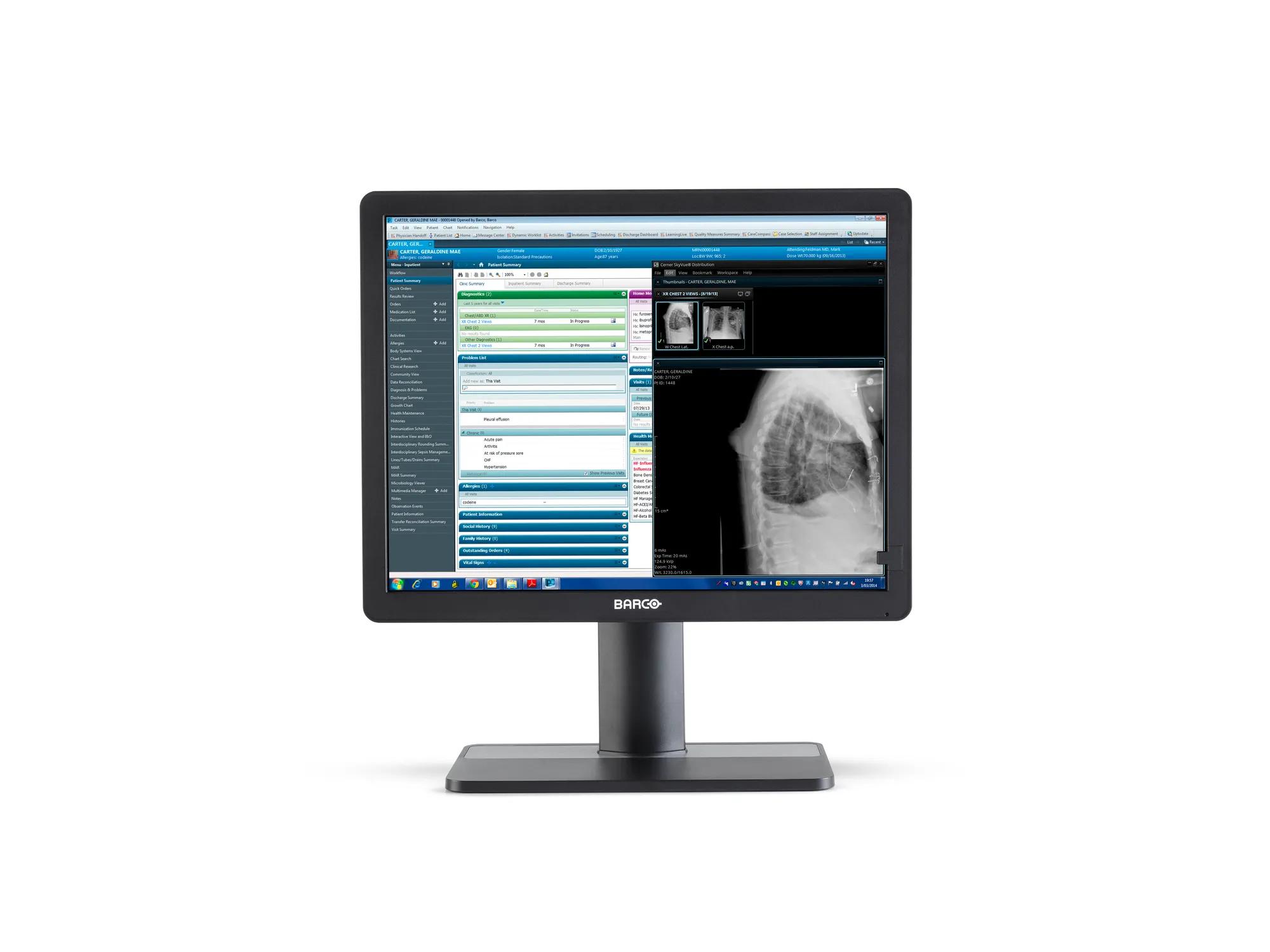
Eonis Color 8MP (MDRC‑8132)
32" clinical display for viewing of medical images and clinical information
- 32" display for viewing medical records
- 8MP screen resolution
- Front sensor for consistent image representation
- Remote QA management tools
- Integrated multimedia functionalities
- Pathology setting for accurate representation of whole slide images
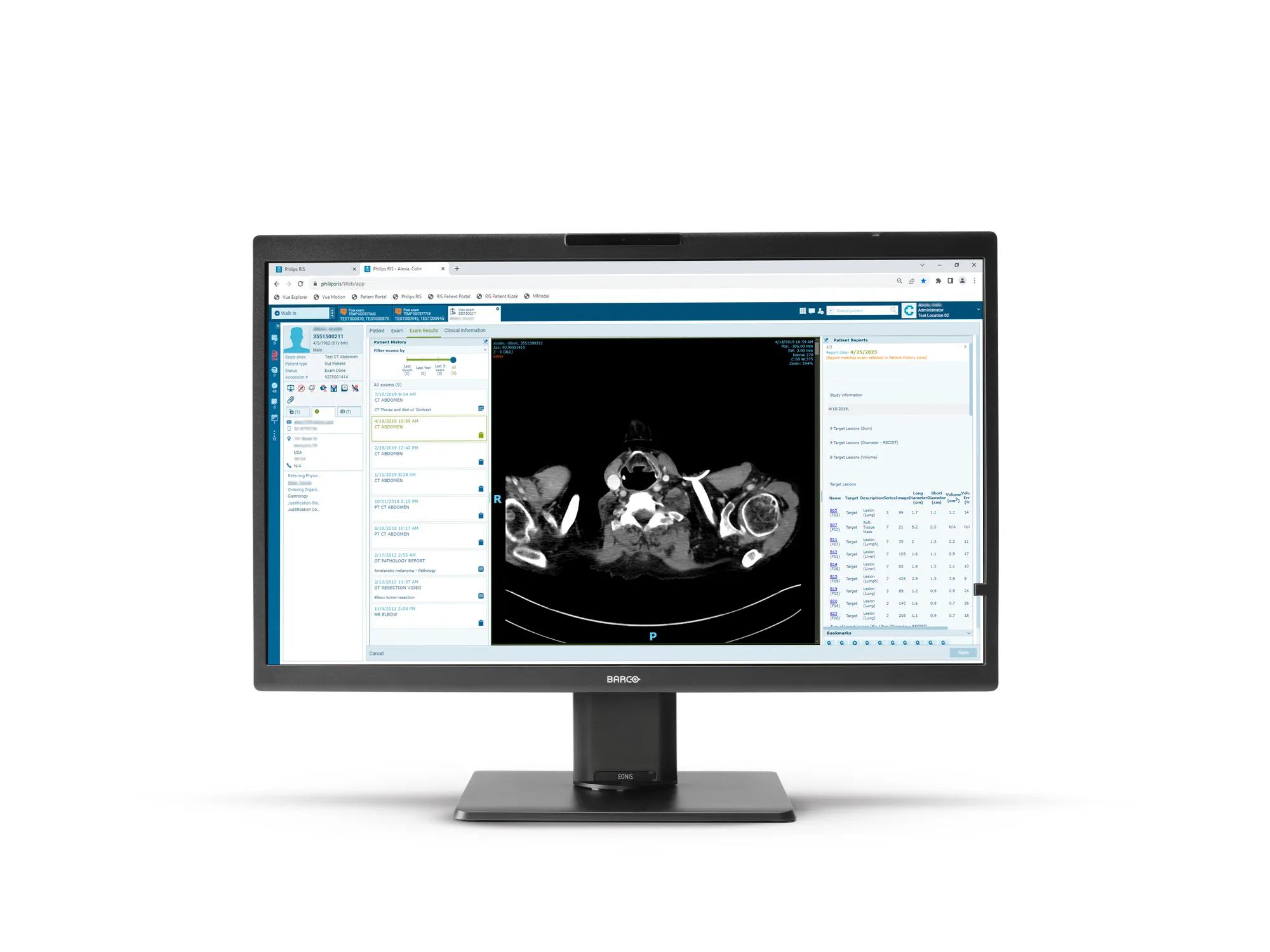
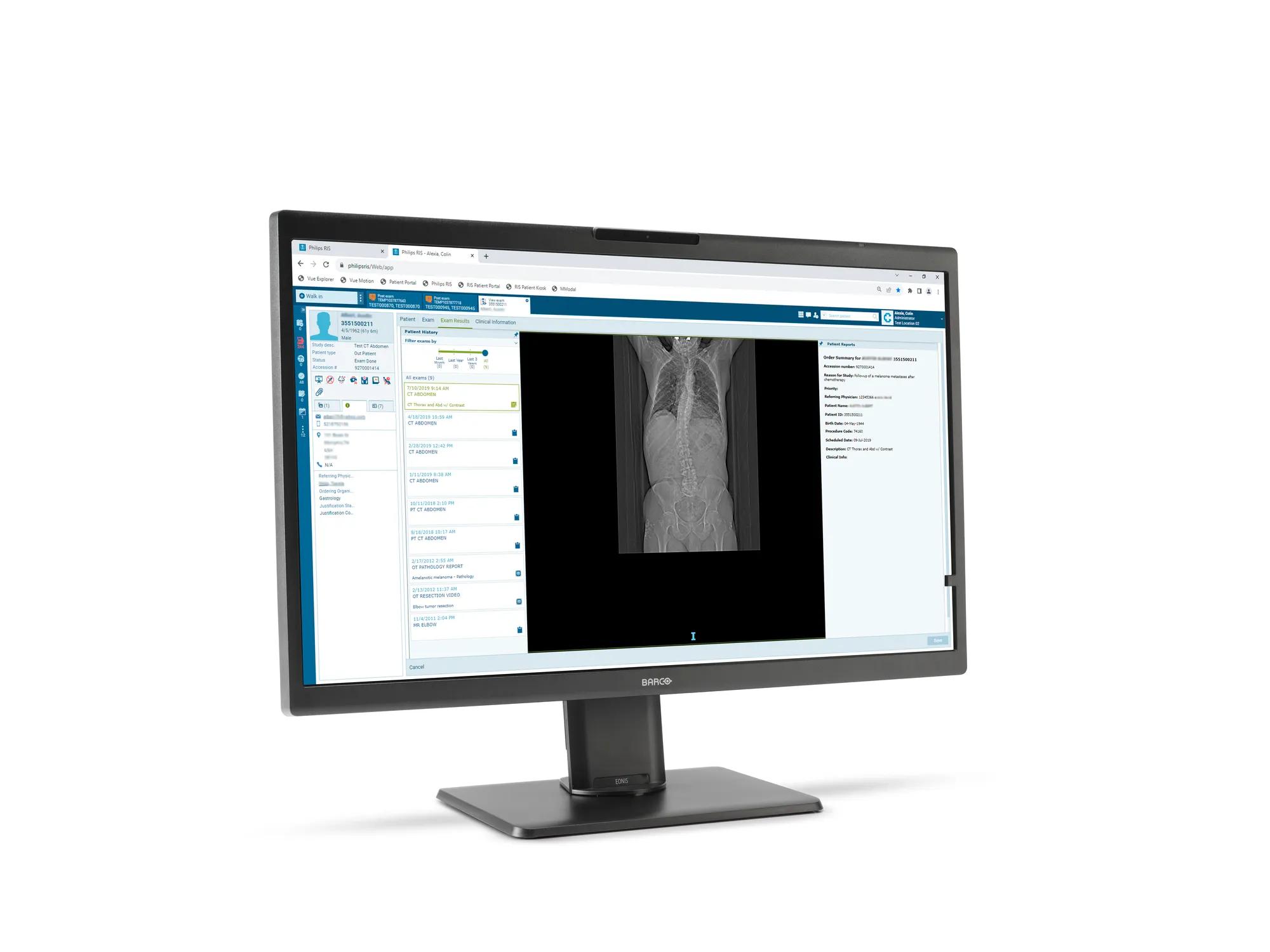
Eonis 8MP is a 32" display built with healthcare specialists in mind. It combines consistent, high image quality and an attractive, versatile design with centralized quality assurance. It also includes built-in multimedia functionalities for easy virtual collaboration.
High image quality
Eonis 8MP offers you 8 megapixels of screen resolution, the highest in our Eonis range. This makes it an ideal choice for healthcare professionals seeking second opinions, emergency or trauma assessments, or referrals, which are not primary diagnoses. Moreover, the display incorporates an innovative front consistency sensor that seamlessly adjusts image quality whenever you power it on.
Additionally, Eonis 8MP has a pathology setting that triggers sRGB color calibration, resulting in excellent images with accurate color representation.
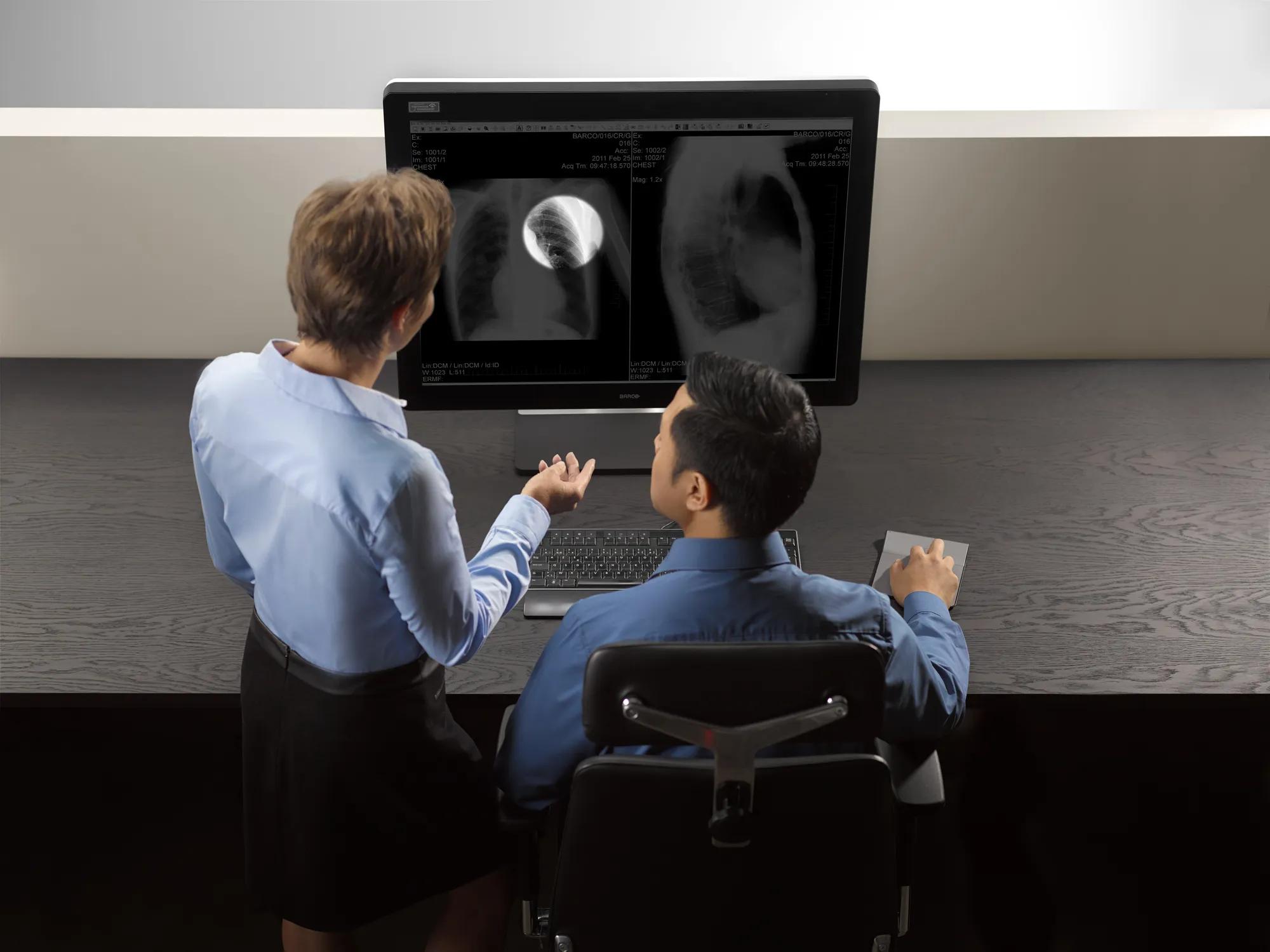
Virtual collaboration was never this easy
The Eonis 8MP extends its capabilities beyond its core functions by providing multimedia features. These streamline intra-hospital and virtual communication among healthcare practitioners, discussing a patient's medical history, electronic medical records and clinical images.
- Built-in camera, microphone, and speakers
- Consistent images and centralized quality assurance
- KVM for seamless switching between workstations
These features promote effective communication across locations and clinical disciplines, making Eonis 8MP an excellent tool for teleconsultations with patients as well.
Centralized quality assurance
Eonis monitors are bundled with Barco's cloud-based QAWeb Enterprise software, an online platform for automated calibration, quality assurance, and asset management. Praised in hospitals around the world, QAWeb Enterprise allows healthcare IT and PACS administrators to centrally and remotely manage image quality across the healthcare organization, at the touch of a button.
A+ ecolabel for Eonis Color 8MP
The Eonis Color 8MP has been subjected to Barco’s ecoscoring protocol and has received an A+ rating. Some key factors that contributed to this rating are:
- Automatic standby mode when device is not in use
- Use of plastics containing 30% recycled content >50% halogen-free cables & >65% halogen-free PCBs
- >90% recyclable packaging
- Unpainted housing to make recycling easier
Specifications
Download spec sheetGeneral Specifications
- Screen technology
- IPS
- Active screen size (diagonal)
- 812.80 mm (32.0")
- Active screen size (H x V)
- 708.48 x 398.52 mm (27.9 x 15.7")
- Aspect ratio (H:V)
- 16:9
- Resolution
- 8MP (3840 x 2160 pixels @ 60 Hz)
- Pixel pitch
- 0.1845 mm
- Color imaging
- Yes
- Gray imaging
- Yes
- Bit depth
- 30 Bit
- Viewing angle (H, V)
- 170°
- Pathology Setting
- Yes
- Color gamut NTSC
- 80.3%
- Color gamut sRGB
- 112%
- sRGB Delta E2000 (typical)
- < 3 (average)
< 5 (maximum)
- RapidFrame
- Yes
- Ambient light presets
- Yes, reading room selection
- Front sensor
- Yes, Front Consistency Sensor
- Maximum luminance (panel typical)
- 500 cd/m²
- DICOM calibrated luminance
- 300 cd/m²
- Contrast ratio (panel typical)
- 1000:1
- Response time ((Tr + Tf)/2) (typical)
- 9.8 ms
- Housing color
- Black (RAL 9004) / White (RAL 9003)
- Video input signals
- 2x DisplayPort 1.4
- Video output signals
- 1x DisplayPort (MST)
- USB ports
- 2x USB-B 2.0 upstream (endpoint)
5x USB-A 2.0 downstream (of which 1 charge port)
- KVM switch
- Yes
- Power rating
- 24 Vdc, 8.3 A
- Power requirements
- This device shall only be powered by the medical approved power supply of Adapter Tech., type ATM200T-P240:
- Input: 100-240 Vac, 50-60 Hz, 2.5-0.9 A
- Output: 24 Vdc, 8.3 A
- Power consumption
- 45.5 W (nominal)
< 0.35 W (hibernate)
< 0.35 W (switched off)
- Dimensions with stand (W x H x D)
- 743 x 518~618 x 238 mm
- Dimensions w/o stand (W x H x D)
- 743 x 459 x 63 mm
- Dimensions packaged (W x H x D)
- 898 x 752 x 358 mm
- Net weight with stand
- 13 kg
- Net weight w/o stand
- 8.4 kg
- Net weight packaged
- 21 kg (without optional accessories)
- Tilt
- -5° to +25°
- Swivel
- -30° to +30°
- Pivot
- N/A
- Height adjustment range
- 100 mm
- Mounting standard
- VESA (100 mm)
- Screen protection
- N/A
- Recommended modalities
- All digital images, except digital mammography.
- Certifications
- CE (Medical Device)
FDA Class I, 510(k) exempt
CCC (China), KC (Korea), INMETRO (Brazil), BIS (India), EAC (Russia) (Pending), UKCA (UK)
Safety specific:
IEC 60950-1:2005+A1:2009+A2:2013
EN 60950-1:2006+A1:2010+A11:2009+A12:2011+A2:2013
IEC 62368-1:2018
EN IEC 62368-1:2020+A11:2020
IEC 60601-1:2005+A1:2012+A2:2020
EN 60601-1:2006+A1:2013+A12:2014+A2:2021
AAMI ES 60601-1:2005+A1:2012+A2:2021
CAN/CSA C22.2 No. 60601-1:2014 (Reaffirmed 2022)
EMI specific:
IEC 60601-1-2:2014+A1:2020 (Ed.4.1)
EN 60601-1-2:2015+A1:2021 (Ed.4.1)
FCC part 15 Class B
ICES-001 Level B
VCCI (Japan)
Environmental:
EU RoHS, China RoHS, REACH, Canada Health, WEEE, Packaging Directive
- Supplied accessories
- User Guide
Quick Install Sheet
Documentation disc
System sheet
Video cables
Mains cables
USB cable
External power supply
- Optional accessories
- Display controller
Touch pad
- QA software
- QAWeb Enterprise
- Warranty
- 3 years
- Operating temperature
- 0 °C to 35 °C (20 °C to 30 °C within specs)
- Storage temperature
- -20 °C to +60 °C
- Operating humidity
- 8 % to 80 % (non-condensing)
- Storage humidity
- 5 % to 85 % (non-condensing)
- Operating pressure
- 70 kPa
- Storage pressure
- 50 to 106 kPa
You can now find all media, brochures, presentations, whitepapers & marketing downloads in our new & improved download center
Visit Media CenterSimilar products
Looking for technical documents or product support?
For technical downloads such as drivers, firmware, manuals, drawings & documentation we would kindly like to direct you to our product support page.
Go to product support