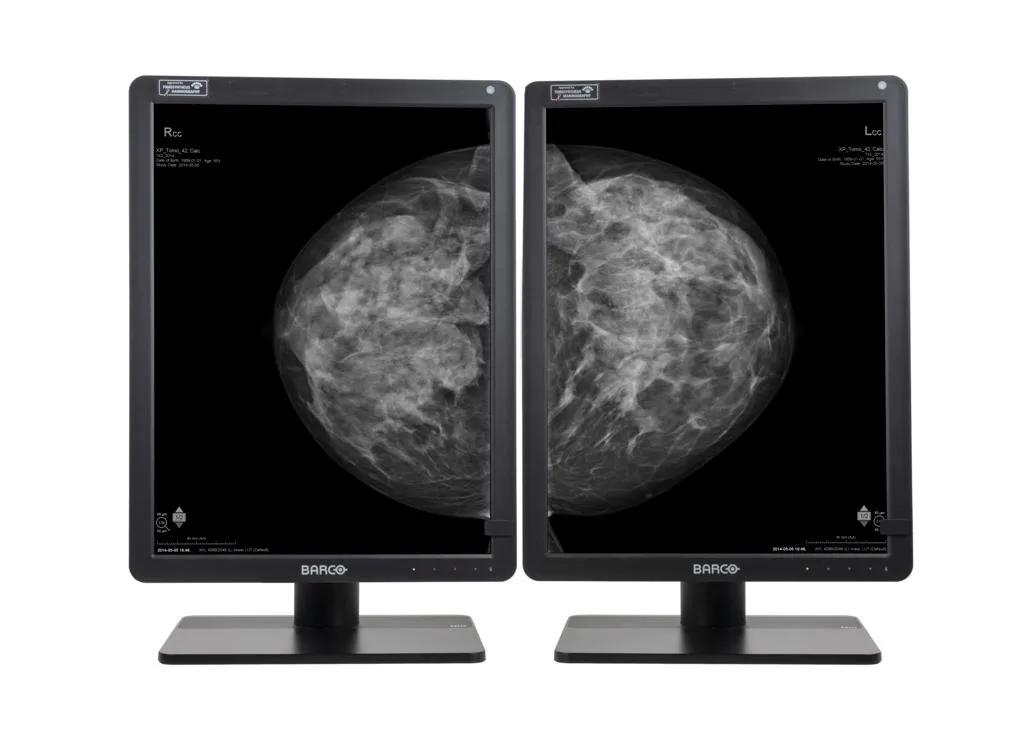
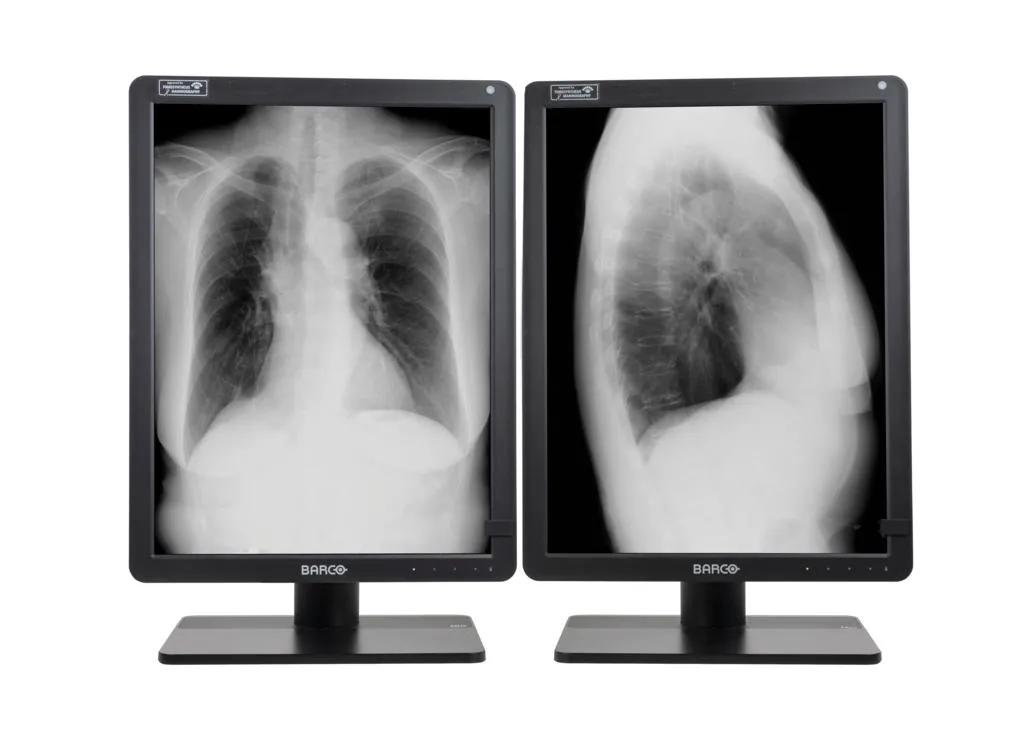
Nio Gray 5.8MP (MDNG‑6221)
5.8MP high-bright grayscale display
- Designed for radiology and 2D/3D mammography
- High-bright, adjustable tint grayscales
- I-Guard and QAWeb Enterprise for always-on compliance
Are you a radiologist specializing in reading breast cases in mainly grayscale modalities, such as 2D mammography and 3D tomosynthesis? If the answer is yes, then the Nio Gray 5.8MP is cut out just for you. It’s fully up to date with capabilities and innovations for today’s breast imaging, making this a modern display system that is efficient and easy for you to use. Furthermore, it’s also perfectly suited for reading grayscale general radiology images in a comfortably high resolution.
Bigger image, more details
Why 5.8MP? Well, in contrast to conventional 5.2MP display systems, you get 12% more pixels on your screen, which means that you can see more details at any given moment. Combine this with the tall 4:3 aspect ratio, which offers more room to view images in their entirety, and you get an ideal combo to start cutting back on additional panning and zooming.
Reliable reading
The Nio Gray 5.8MP offers you more Just Noticeable Differences, thanks to its high brightness and contrast ratio. Our integrated stability, calibration and uniformity technologies make sure that image quality, light output and DICOM compliance remain consistent over the years.
Efficient workflow
The Nio Gray 5.8MP is more than a grayscale monitor alone. It offers many ways to personalize settings to your liking, such as preferred tints of white or viewing angle.
On top of that, it can help you improve your efficiency and speed, thanks to the set of Intuitive Workflow Tools included with our MXRT medical display controllers.
Did you know that SpotView, for example, makes it possible to make an area you choose twice as bright as it was originally? It’s been proven to help radiologists reduce their reading time by as much as 15.5%. You can also define user profiles, saving you and your colleagues time. These automatically adjust the display in situations where it is shared by multiple people.
Long lifetime, clear view
The Nio Gray 5.8MP adheres to the latest medical standards for medical devices in Europe & MQSA in the United States. It’s also easy to install our free and highly secured QAWeb Enterprise application, with which you can rely onintervention-free, remote quality assurance. The tool makes it easy for PACS managers to centrally manage their Barco displays’ uptime and compliance – anytime, anywhere.
To summarize, your Nio Gray 5.8MP monitor is a functional, easy-to-use diagnostic display system, fully up to date with today’s innovations in general grayscale radiology, as well as 2D and 3D mammography. It comes with a 5-year warranty on its all components.
Ensuring diagnostic confidence with MDR Class IIa
Our radiology displays are MDR-certified as Class IIa. Their product information has been reviewed and cleared by independent medical and technical experts, and is audited yearly. In other words, we ensure diagnostic confidence and peace of mind for our users.
Technologies that enhance image quality:
- More detail on your screen, with 5.8MP resolution
- Designed to show breast images entirely, with 3:4 aspect ratio
- Increased contrast, with 1400:1 contrast ratio and 600 to 1000 cd/m² calibrated luminance
- Consistent brightness and grays, with Uniform Luminance Technology and SteadyGray
- Always stable DICOM images and auto QA, with I-Guard front sensor and optionally, QAWeb Enterprise
- Possibility to boost luminance, with I-Luminate and SpotView
- Optional settings and tools to adjust the monitor to your workflow, with Intuitive Workflow Tools
A ecolabel for Nio Gray 5.8MP
The Nio Gray 5.8MP has been subjected to Barco’s ecoscoring protocol and has received an A rating. Some key factors that contributed to this rating are:
- Energy efficient power supply, energy efficient standby and off modes
- Possibility to automatically switch to standby mode when device is not in use
- Halogen-free cables and plastics
- Use of recycled cardboard in packaging (>85% recycled content)
- Product design optimized for disassembly with common tools
Specifications
Download spec sheetGeneral Specifications
- Screen technology
- LCD
- Active screen size (diagonal)
- 541 mm (21.3")
- Active screen size (H x V)
- 324 x 433 mm (12.77" x 17")
- Aspect ratio (H:V)
- 3:4 for each display in portrait mode, 3:2 overall
- Resolution
- 5.8 MP (2100 x 2800 pixels)
- Pixel pitch
- 0.1545 mm
- Gray imaging
- Yes
- Bit depth
- 10 bit
- Viewing angle (H, V)
- 178°
- Optical glass
- MDNG-6221 option SPEF: Yes
MDNG-6221 option SNEF: No
- Uniformity correction
- ULT
- SteadyGray
- Yes
- Ambient light presets
- Yes, reading room selection
- I-Luminate
- Yes
- Ambient light sensor
- MDNG-6221 option SPEF: Yes MDNG-6221 option SNEF: No
- Front sensor
- Yes
- Maximum luminance
- 1300 cd/m² (MDNG-6221 SNEF / SPEF)
650 cd/m² (MDNG-6221 NC EM)
- DICOM calibrated luminance
- [MDNG-6221 SNEF / SPEF]
Warrantied maximum 1000 cd/m²
Factory default 600 cd/m²
[MDNG-6221 NC EM]
500 cd/m²
- Contrast ratio (panel typical)
- 1400:1
- Response time ((Tr + Tf)/2) (typical)
- 12.5 ms
- Housing color
- RAL 9003 / RAL 9004
- Video input signals
- DVI-D Dual Link (2x), DisplayPort(2x)
- USB ports
- 1x USB 2.0 upstream (endpoint)
2x USB 2.0 downstream
- Power rating
- 24 Vdc, 5 A; 5 Vdc, 0.1 A
- Power requirements
- This device shall only be powered by the medical approved power supply:
Adapter Technology, type CMD160-P240:
- Input: 100-240 Vac, 50/60 Hz, 1.9-0.8 A
- Output: 24 Vdc, 6.3 A; 5 Vdc, 0.5 A
- Dimensions with stand (W x H x D)
- Portrait: 378 x 528~628 x 235 mm
Landscape:491 x 472~572 x 235 mm
- Dimensions w/o stand (W x H x D)
- Portrait: 378 x 491 x 81 mm
Landscape:491 x 378 x 81 mm
- Dimensions packaged (W x H x D)
- 500 x 280 x 670 mm
- Net weight with stand
- [MDNG-6221 SPEF]
11.6 kg
[MDNG-6221 SNEF / NC EM]
10.2 kg
- Net weight w/o stand
- [MDNG-6221 SPEF]
6.6 kg
[MDNG-6221 SNEF / NC EM]
5.2 kg
- Net weight packaged
- [MDNG-6221 SPEF]
17 kg (without optional accessories)
[MDNG-6221 SNEF / NC EM]
15.7 kg (without optional accessories)
- Tilt
- -10° to 30°
- Swivel
- -45° to +45°
- Pivot
- 90°
- Height adjustment range
- 100 mm
- Mounting standard
- VESA (100 mm)
- Screen protection
- [MDNG-6221 SPEF]
Hier wat tekst
- Available: yes
- Anti-Reflective coating: yes
- Material: glass
- Available: no
- Recommended modalities
- All digital images, including digital mammography and breast tomosynthesis.
- Certifications
- [MDNG-6221 SNEF / SPEF]
CE0123 (Medical Device)
FDA 510(K) K170476
CCC (China), KC (Korea), INMETRO (Brazil - Product Numbers: K9300370B, K9300372B), BIS (India), EAC (Russia, Kazakhstan, Belarus, Armenia and Kyrgyzstan)
Safety specific:
IEC 60950-1:2005+A1:2009+A2:2013
EN 60950-1:2006+A1:2010+A11:2009+A12:2011+A2:2013
IEC 62368-1:2018
EN IEC 62368-1:2020+A11:2020
IEC 60601-1:2005+A1:2012+A2:2020
EN 60601-1:2006+A1:2013+A12:2014+A2:2021
AAMI ES 60601-1:2005+A1:2012+A2:2021
CAN/CSA C22.2 No. 60601-1:2014 (Reaffirmed 2022)
EMI specific:
IEC 60601-1-2:2014+A1:2020 (Ed.4.1)
EN 60601-1-2:2015+A1:2021 (Ed.4.1)
FCC part 15 Class B
ICES-001 Level B
VCCI (Japan)
Environmental:
EU RoHS, China RoHS, REACH, Canada Health
WEEE, Packaging Directive
[MDNG-6221 NC EM]
CE0123 (Medical Device)
INMETRO (Brazil - Product Numbers: K9300371B), BIS (India)
Safety specific:
IEC 60950-1:2005+A1:2009+A2:2013
EN 60950-1:2006+A1:2010+A11:2009+A12:2011+A2:2013
IEC 62368-1:2018
EN IEC 62368-1:2020+A11:2020
IEC 60601-1:2005+A1:2012+A2:2020
EN 60601-1:2006+A1:2013+A12:2014+A2:2021
AAMI ES 60601-1:2005+A1:2012+A2:2021
CAN/CSA C22.2 No. 60601-1:2014 (Reaffirmed 2022)
EMI specific:
IEC 60601-1-2:2014+A1:2020 (Ed.4.1)
EN 60601-1-2:2015+A1:2021 (Ed.4.1)
Environmental:
EU RoHS, REACH
WEEE, Packaging Directive
- Supplied accessories
- User Guide
- System sheet
- Documentation disc
- Video cable (1x DisplayPort)
- Mains cable(s)
- USB 2.0 cable
- External power supply
- Optional accessories
- Display controller
- QA software
- QAWeb
- Warranty
- [MDNG-6221 SNEF / SPEF]
5 years, including 40000 hours backlight warranty
[MDNG-6221 NC EM]
3 years, including 20000 hours backlight warranty
- Operating temperature
- 0 °C to 40 °C (15 °C to 30 °C within specs)
- Storage temperature
- -20 °C to 60 °C
- Operating humidity
- 8 % to 80 % (non-condensing)
- Storage humidity
- 5 % to 85 % (non-condensing)
- Operating pressure
- 70 kPa
- Storage pressure
- 50 to 106 kPa
EssentialCare for Diagnostic imaging
Protect your clinical efficiency
- Assure your display's quality and compliance
- Benefit from proactive, data driven support
- Avoid downtime with upfront replacements

ConnectCare for Diagnostic imaging
Get the very best out of your investment
- Extend your diagnostic display fleet's lifetime and enjoy optimal performance
- Expert support in monitoring & optimizing your fleet
- Strategic insights to maximize your diagnostic display investment

You can now find all media, brochures, presentations, whitepapers & marketing downloads in our new & improved download center
Visit Media CenterLooking for technical documents or product support?
For technical downloads such as drivers, firmware, manuals, drawings & documentation we would kindly like to direct you to our product support page.
Go to product support